BDyn
BDyn: sistema de estabilización dinámica posterior certificado por el MDR de la UE
BDyn
BDyn: sistema de estabilización dinámica posterior certificado por el MDR de la UE
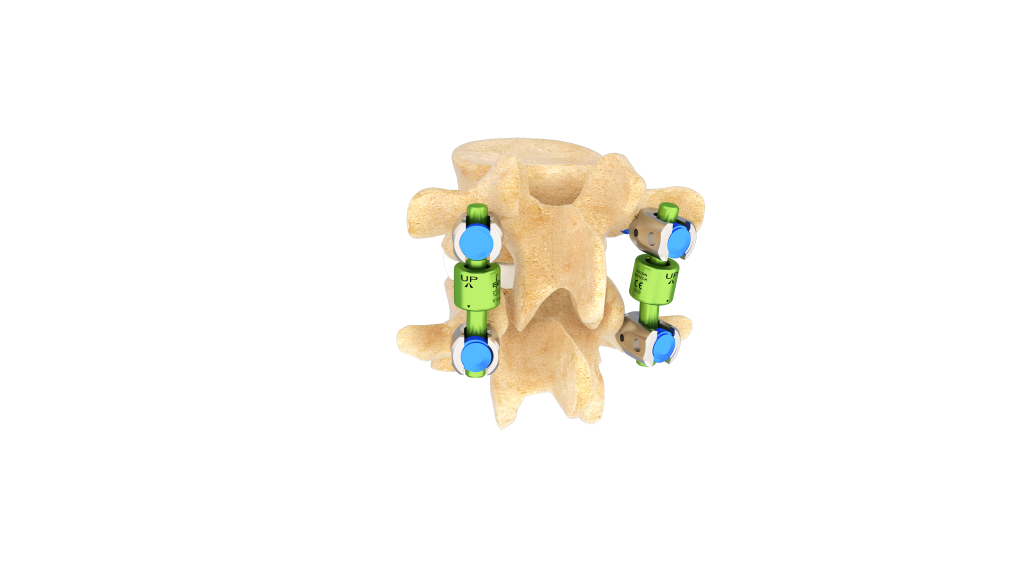
El dispositivo BDyn ofrece una solución para estabilización dinámica posterior de la columna vertebral, certificada por el MDR de la UE, diseñada para los niveles desde T10 hasta el sacro S1.
Desarrollado para tratar determinadas afecciones degenerativas lumbares, BDyn proporciona una estabilización controlada al mientras sigue permitiendo movimiento.
*Guérin et al. – Einfluss eines dynamischen Stabilisierungssystems auf die segmentale Bewegung, den Bandscheibendruck und die Kontaktkräfte der Facettengelenke. Eine biomechanische In-vitro-Studie
- un cojín de silicona que absorbe la carga y reduce la presión sobre el disco intervertebral y las articulaciones facetarias,
- un anillo de elastómero que limita la hipermovilidad y controla la extensión segmentaria.
- Patología degenerativa del disco intervertebral
- Estenosis de canal
- Espondilolistesis degenerativa de grado 1
- Inestabilidad segmentaria
MATERIAL
- Titanio
- Silícona
- PCU (Uretano De Policarbonato)
BDyn implant is a class III medical device. The CE conformity has been carried out by the notified body SGS Belgium NV (CE1639).The BDyn instrument kit is composed of class Ir instruments medical devices manufactured by COUSIN BIOTECH S.A.S & Neuro France Implants. The management system of COUSIN BIOTECH S.A.S is certified for compliance with ISO 13485 standard. Please read carefully the instructions for use before using the device. The IFU is available electronically at: https://www.cousin-biotech.com/en/implant-notices.
Cousin Biotech is the legal manufacturer of the medical devices proposed by Cousin Surgery.