BDyn
Posterior Dynamic Stabilization System
BDyn
Posterior Dynamic Stabilization System
BDyn – EU MDR-Certified Posterior Dynamic Stabilization System
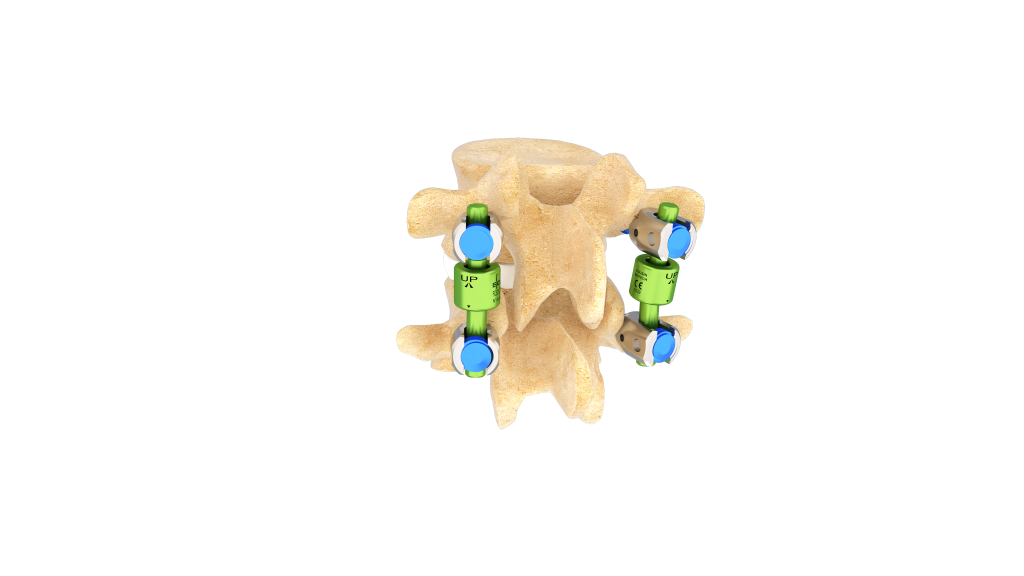
The BDyn device offers a solution for posterior dynamic spinal stabilization, certified under the EU MDR, designed for levels from T10 to sacrum S1.
Developed to treat certain lumbar degenerative conditions, BDyn provides controlled stabilization while preserving motion*.
*Guérin and al. – Effect of dynamic stabilization system on the segmental motion, intradiscal pressure and facet joint contact forces. An in vitro biomechanical study
- a silicone cushion that absorbs load and reduces stress on the intervertebral disc and facet joints,
- an elastomer ring that limits hypermobility and controls segmental extension.
- Degenerative intervertebral disc disease
- Spinal canal stenosis
- Degenerative spondylolisthesis grade 1
- Segmental instability
MATERIAL
- Titanium
- Silicone
- PCU (Polycarbonate Urethane)
BDyn implant is a class III medical device. The CE conformity has been carried out by the notified body SGS Belgium NV (CE1639).
The BDyn instrument kit is composed of class Ir instruments medical devices manufactured by COUSIN BIOTECH S.A.S & Neuro France Implants. The management system of COUSIN BIOTECH S.A.S is certified for compliance with ISO 13485 standard. Please read carefully the instructions for use before using the device. The IFU is available electronically at: https://www.cousin-biotech.com/en/implant-notices
Cousin Biotech is the legal manufacturer of the medical devices proposed by Cousin Surgery.